Blood Tests Could Provide Early Warning of Alzheimers Disease
Alzheimer’s disease remains a frustratingly difficult condition to manage for the millions of patients affected worldwide and their families. The cause of the disease is still not properly understood, and by the time memory loss and cognitive decline become apparent, the underlying brain pathology has often been quietly building for decades.
Soon, though it may be possible to diagnose impending Alzheimer’s disease ahead of time, before symptoms have taken hold. New research suggests this could be achieved through a simple blood draw, providing clinicians and patients precious time to manage the condition and plan ahead.
Early Warning

A hallmark of Alzheimer’s disease is the buildup of amyloid and tau protein in the brain. Despite decades of research, the protein’s precise role in the disease remains somewhat unclear. Typically, Alzheimer’s disease is diagnosed by symptoms like memory loss and cognitive decline, with later investigation revealing the presence of elevated levels of these proteins in the brain. This investigation often involves brain imaging or invasive and painful spinal fluid tests.
However, researchers have developed a new predictive model that suggests it’s possible to estimate when a person will begin showing Alzheimer’s symptoms, based on a similar marker. The key ingredient is another protein called p-tau217, which circulates in the blood plasma and mirrors the slow, steady accumulation of amyloid and tau protein within the brain tissues.
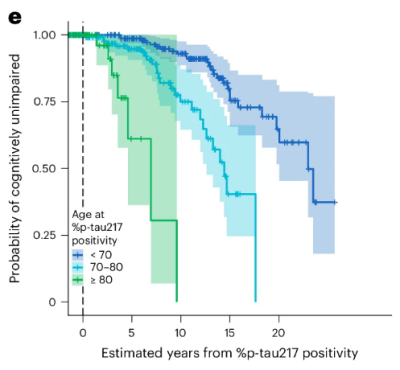
Earlier research had previously established that p-tau217 levels tended to track alongside the rise of amyloid and tau buildup in the brain. This indicated that the level of p-tau217 in the blood could be a proxy for how far along the disease process has progressed. This allows the estimation of how many years remain before symptoms emerge for a given patient, with the research study suggesting this could be as specific as a margin of three or four years.
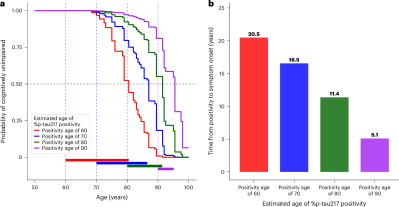
The research study drew on data from 603 older adults enrolled in two long-running studies — the Knight Alzheimer Disease Research Center at WashU Medicine and the multi-site Alzheimer’s Disease Neuroimaging Initiative. Plasma p-tau217 was measured using simple blood draws. One of the more interesting findings concerns age. The model revealed that the interval between elevated p-tau217 levels and symptom onset isn’t fixed. A person whose levels first ticked upwards at the age 60 might not develop noticeable cognitive problems for another two decades. But if that same elevation appeared at age 80, symptoms tended to follow within about eleven years. It suggests that younger brains appear to tolerate Alzheimer’s-related pathology for longer, while older brains are less able to withstand the protein buildup once the process gets underway.
Right now, p-tau217 testing is primarily used to help confirm an Alzheimer’s diagnosis in patients who are already experiencing cognitive trouble. It isn’t recommended for screening asymptomatic individuals outside of research settings. However, using p-tau217 as a predictive marker has obvious potential. If clinical trials for preventive Alzheimer’s drugs could enroll participants based on a predicted timeline for symptom onset, rather than waiting years for decline to actually happen, those trials could become dramatically shorter and cheaper to run, and more efficient in general. Perhaps more importantly, it has the potential to give patients a better understanding of what lies ahead, allowing them to plan ahead before cognitive symptoms become an unmanageable imposition on their life.
The research team has made their model development code publicly available on Github and created a web-based tool that lets other researchers explore the clock models in detail. Looking ahead, they note that combining p-tau217 with other blood-based biomarkers linked to cognitive decline could sharpen predictions even further. It’s still early days, but a future where a routine blood test could serve as an early-warning system for Alzheimer’s is looking more plausible than it did a few years ago.


